Longitudinal Diffusion from Principles and Practice of Chromatography
Principles and Practice of Chromatography
by Raymond P. W. Scott,
part of the Chrom-Ed Series
Longitudinal Diffusion
Solutes when contained in a fluid naturally diffuse and spread driven by their concentration gradient. Thus, in a chromatographic column a discrete solute band will diffuse in the gas or liquid mobile phase. It also follows, that because the diffusion process is random in nature, a concentration curve that is Gaussian in form will be produced. This diffusion effect occurs in the mobile phase of both GC and LC columns. The diffusion process is depicted in figure 21.
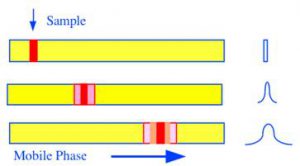
Figure 21 Peak Dispersion by Longitudinal Diffusion
The longer the solute band remains in the column, the greater will be the extent of diffusion. The time the solute remains in the column is inversely proportional to the mobile phase velocity, so, the dispersion will also be inversely proportional to the mobile phase velocity. Van Deemter et al. derived the following expression for the variance contribution by longitudinal diffusion, (), to the overall variance/unit length of the column.
![]() (5)
(5)
where (Dm) is the diffusivity of the solute in the mobile phase,
(u) is the linear velocity of the mobile phase,
and (g) is a constant that depended on the quality of the packing.
The Resistance to Mass Transfer in the Mobile Phase
During passage through a chromatographic column, the solute molecules are constantly and reversibly transferring from the mobile phase to the stationary phase. This transfer is not instantaneous; time is required for the molecules to pass (by diffusion) through the mobile phase to reach the interface and enter the stationary phase. Those molecules close to the stationary phase enter it immediately, whereas those molecules some distance away will find their way to it some time later. Since the mobile phase is continually moving, during this time interval, those molecules that remain in the mobile phase will be swept along the column and dispersed away from those molecules that were close and entered the stationary phase immediately. This process is depicted in figure 22. The diagram shows 6 solute molecules in the mobile phase and the pair closest to the surface, (1 and 2), enter the stationary phase immediately. While molecules 3 and 4 diffuse through the mobile phase to the interface, the mobile phase moves on. As a consequence, when molecules 3 and 4 reach the interface, they enter the stationary phase some distance ahead of the first two. Finally, while molecules 5 and 6 diffuse to the interface, the mobile phase has moved even further down the column until molecules 5 and 6 enter the stationary phase ahead of molecules 3 and 4. Thus, the 6 molecules, originally relatively close together, are now spread out in the stationary phase. This explanation, although over-simplified, gives a correct description of the mechanism of mass transfer dispersion.
