Hydrocarbon Analysis
Due to the perceived toxicity and carcinogenic character of the aromatic hydrocarbons, the presence of these materials is carefully monitored in all areas where they might enter the human food chain. The analysis of water for aromatic hydrocarbons, particularly surface water in those areas where contamination might take place, is a common assay made by the public analyst. It is essential to be able to measure concentrations in the ppb levels, and thus GC method employing a high sensitive detector is essential. Nevertheless, even if a high sensitivity detector is employed, some sample concentration will be necessary to measure contaminants at such low levels.
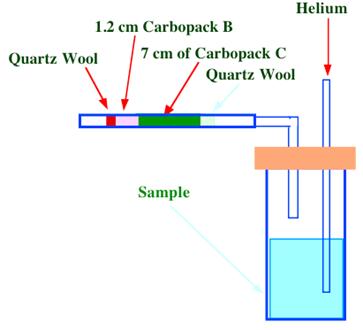
Figure 37. Purge and Trap System for Aromatic Hydrocarbons in Water
One method is the purge and trap procedure, using a solid adsorbent to remove the hydrocarbon vapors. A diagram of the purge and trap system is shown in figure 37. A 5 ml sample of spiked water was placed in a small vial through which a stream of helium was passed at a flow rate of 40 ml/min. The purge was carried out at room temperature for 11 min. After bubbling through the sample, the helium passed through a stainless steel adsorption tube, 1/8 in. diameter having a 7 cm length packed with Carbopack C and a 1.2 cm length packed with Carbopack B. Carbopack C is 20/40 mesh, graphitized carbon, having a surface area of 10 m2/g and the short length of Carbopack B packing had a surface area of about 100 m2/g. The short length of packing with the higher surface area (thus high adsorptive capacity) was to used to ensure that none of the sample material was eluted through the adsorption bed and lost. The sample vial was then removed and the trap purged dry with 40 ml/min. of dry helium for 5 min. The trap was then transferred to a small heating oven and the contents desorbed onto the column at 250°C with a 4 min. bake at 260°C. A chromatogram of the separation that was obtained is shown in figure 38.
